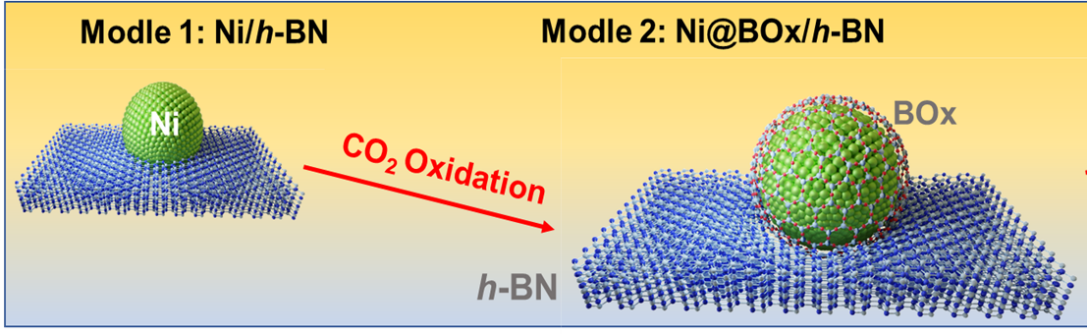
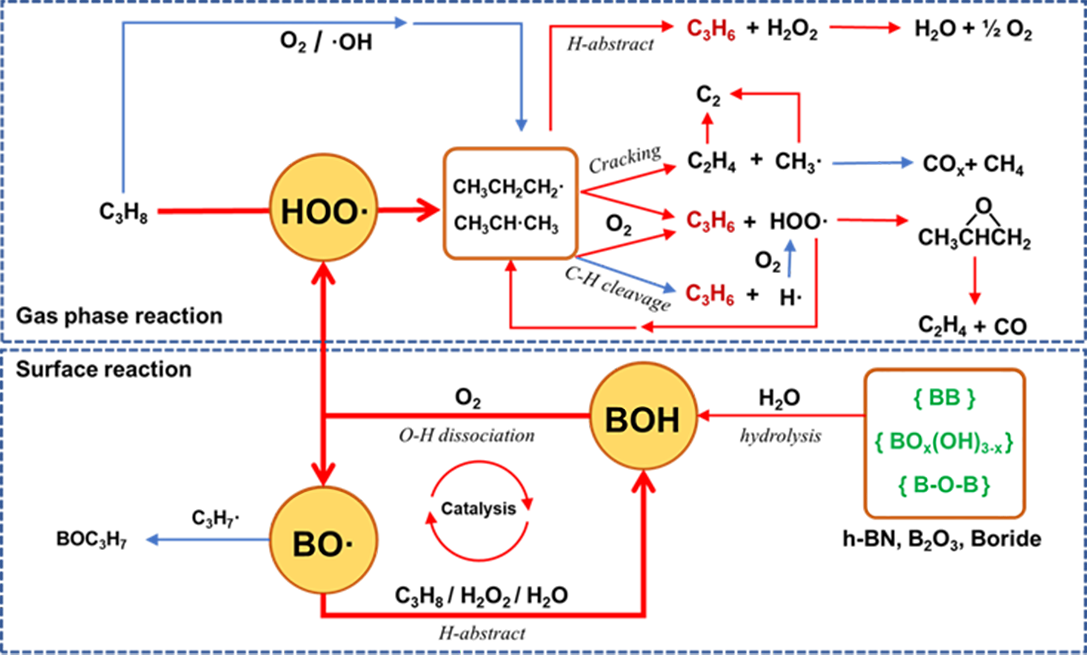
Propene is one of the most important chemical feedstocks and building blocks of polymers. The oxidative dehydrogenation of propane (ODHP, C3H8+1/2O2→C3H6+H2O) is a promising pathway to synthesize propene, as it is an exothermic reaction with no thermodynamic limitation and prevents the frequent carbon deposition removal and catalyst regeneration that occur during the commercialized propane direct dehydrogenation process (PDH). Our aim is developing advanced heterogeneous catalyst to enable the low-temperature efficient, selective and stable oxidative dehydrogenation of propane and other low alkanes to olefins.
By developing the subsurface metal promoter strategy, we managed to construct a generalizable strategy to accelerate the radical initiation activity of boron based catalysts and promotes their low temperature activity and olefin selectivity. The modulation methods and performance descriptors for these systems are also proposed.